Strategy
Back to School 2017: Breach of contract
Why the drug industry must rectify its relationship with society.
Merck & Co. Inc. Chairman and CEO Kenneth Frazier’s resignation in August from a White House advisory council was an act of principle, and a reminder that 30 years ago Merck was one of the most respected companies in America.
Few pharma executives or members of the public remember that former Merck CEO and Chairman Roy Vagelos pledged in 1990 to cap drug price increases at or below the rate of inflation, and later warned that high launch prices could destroy public faith in the industry.
When President Donald Trump fired back with a tweet that Frazier would now have time to “LOWER RIPOFF DRUG PRICES!” he sought to deflect attention from the reasons for Frazier’s resignation to a much more popular theme: disdain for drug companies. Trump knew that most Americans would applaud his attack on drug prices.
Trump’s comfort in using Merck as a shield against the outrage sparked by his response to the violence in Charlottesville shows the dire state of the drug industry’s reputation. And this sorry state of affairs exemplifies the breakdown of the social contract between the drug industry and the society it is supposed to serve.
Why this contract exists, and why the drug industry must repair its relationship with society, is the subject of BioCentury’s 25th Back to School essay.
Patients must believe the companies that create and sell medicines are on their side.
At the most basic level, the social contract starts with the idea that industry’s sole purpose isn’t to sell pills and vials, or to increase shareholder value, but to help patients live better and longer.
When the public believes drug companies are honoring the contract, governments and philanthropies are willing to fund biomedical research that leads to knowledge that industry can translate, via gigantic and risky investments, into medicines. Society will pay enough for medicines to ensure that companies can attract the investment that is required to fund a long, uncertain R&D process. Governments provide strong, science-based regulation and well-defined IP protections with the explicit understanding that monopolies will end at prespecified times. And patients willingly participate in risky clinical trials for the greater good.
These contributions from society and individuals create obligations for drug companies.
Drug developers must make the investments required to produce medicines that help patients improve their health, and in the process pave the way for broad advances in public health and prosperity.
Medical product manufacturers are obliged to abide by science-based regulations, and release their grasp on captive markets when exclusivity periods terminate.
Above all, for the contract to work, patients must have access to medicines that will improve their lives at prices they can afford. And patients must believe the companies that create and sell medicines are on their side.
Judged against these criteria, the drug industry is failing despite its lip service to these obligations.
It would be wrong to suggest drug companies are solely responsible for this debacle. The greatest unmet medical need is access to affordable healthcare, and on this criterion, evidence of the broken social contract is everywhere.
The poor reputation of insurance companies, hospitals and, on some metrics, even healthcare providers, at least in the U.S., reflects the widely held view that the healthcare system is not working in the public’s favor.
Moreover, the drug industry is entitled to shine a light on profits-before-patients behavior by insurance companies, PBMs, hospitals and physicians. They all are culpable in making healthcare unaffordable and inaccessible.
But drug companies are not helping themselves by only trying to deflect the blame for high drug prices and denial of access to the other healthcare players, or by trumpeting ad nauseam that other healthcare expenses account for the vast bulk of spending on public health.
These arguments are irrelevant to the patient who cannot afford a necessary drug.
Anyone who is or knows a patient who is unable to obtain medicines, or who is appalled by the price of the drug they have been able to obtain, has concluded the drug industry is not holding up its end of the deal.
That sense of unfairness is leading politicians around the world to look for ways to squeeze pharma profits and weaken IP protection for the industry’s products.
This year, Back to School contends that drug manufacturers, their managers and their boards of directors must acknowledge they are part of the problem, and change course.
To fulfill the social contract, Back to School argues that drug companies must stop making choices that belie the industry’s claims that it puts patients first.
They must align management incentives with patient and public health, not just shareholder returns.
And most importantly, drug companies must be seen to be improving access to medicines.
Although fulfilling the social contract is almost entirely the responsibility of individual companies, the drug industry collectively needs to agree and adhere to best practices and principles on pricing, access and transparency -- because the reputations of companies that do a good job of meeting their commitments to patients and society are dragged down by those that violate the contract. Some steps can only be accomplished, or will only be equitable, if there is a collective commitment.
Only if industry and its companies take aggressive, visible steps to solve access problems will they be acknowledged for creating new drugs that improve public health. And only then will they have properly earned the right to ask for society’s continued investment in medical innovation and for society to pay for innovative drugs based on the value they create.
The situation
The biopharma industry relies on government policies to fuel innovation, including science-based regulatory systems; strong but finite IP protections; and public investment in scientific research coupled with policies that facilitate uptake of that research by the private sector.
Medical product development also relies heavily on support from individual patients and patient organizations, as participants in risky and often painful clinical research, as advocates for efficient regulatory and compassionate coverage policies, and eventually as consumers.
Giving term-limited monopolies to biopharma innovators for novel medicines, paying premium prices for novel innovative drugs, investing tens of billions of dollars annually for biomedical research and putting individual patient lives on the line in clinical trials represents an enormous investment by society (see “Cutting the Check” & “Footing the Bill”).
In return, society rightfully expects the drug industry to apply its unique abilities to turn scientific advances into drugs that improve individual and public health outcomes.
Over the past three decades, the biopharma industry in fact has delivered extraordinary advances in medicine. These include transforming AIDS from a pandemic killer to a manageable disease and developing vaccines that prevent cervical and other HPV-related cancers.
The idea that a pill can prevent a disease like cystic fibrosis from choking the life out of a child, or banish HCV from someone’s body is nothing short of astounding.
There is evidence that Kymriah tisagenlecleucel from Novartis AG, which on Aug. 30 became the first CAR T cell therapy to gain FDA approval, can cure some leukemia patients whose disease would have been fatal. And industry’s pipeline contains many more potential cures.
Ironically, the more successful the drug industry has been in producing important medicines, the lower it has sunk in public perception.
Anyone who is or knows a patient who is unable to obtain medicines has concluded the drug industry is not holding up its end of the deal.
According to a September 2016 Kaiser Family Foundation poll, only 56% of Americans believe that prescription drugs developed over the past 20 years have made the lives of people in the U.S. better. That’s down from 73% in March 2008.
The 2016 Harris Poll Study of Reputation Equity and Risk Across the Health Care Sector paints a worse picture. Pharmaceutical and biotech companies are in last or second-to-last place compared with hospitals, providers and insurers on reputation, putting patients first, making a positive difference and socially responsible behavior (see “Opinion Poll”).
The drug industry’s huge achievements have been drowned out by reports that in the U.S. some patients buy groceries for their families instead of picking up their insulin. That women and men hobbled by rheumatoid arthritis forgo biologic drugs that could make their lives tolerable. And that leukemia patients whose lives were saved by a tyrosine kinase inhibitor gamble that the cancer will stay in remission if they skip their pills for a few weeks.
In other parts of the world, many patients don’t get as far as a pharmacy. In rich countries, governments routinely decide it’s simply too expensive to offer their citizens some drugs that enhance and save lives. In poor countries, ordinary people don’t consider purchasing medicines that people in the U.S., Europe and Japan wouldn’t think of doing without.
The fact that most drug companies live up to most of their commitments to society most of the time is not good enough: partial success is perceived as total failure.
“The fact is that too many people can’t afford the medicines that they need,” FDA Commissioner Scott Gottlieb told Congress in July.
Figure: Footing the bill
National governments are among the largest purchasers of prescription pharmaceuticals, with five of the biggest developed nations spending almost $300 billion annually from 2012 to 2014. Data below include government spending on prescription pharmaceuticals across all hospital, outpatient and other channels, except as noted. Values are nominal and have been converted to U.S. dollars using the exchange rate on the last day of the country’s fiscal year. (A) Retail prescription sales only; (B) Fiscal year is from April 1 to March 31; (C) Fiscal year is from April 1 to March 31, and data for FY10, FY11 and FY15 not available; $B; Sources: CMS, NHS Digital, Eurostat, French Health Accounts Committee, Japanese Ministry of Health, Labour and Welfare (MHLW)
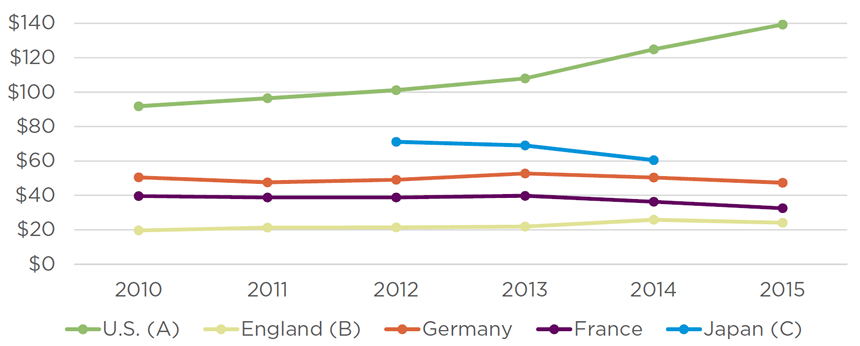
Figure: Opinion poll
The 2016 Harris Poll Study of Reputation Equity and Risk Across the Health Care Sector asked 1,018 U.S. adults about their opinions of pharmaceutical and biotechnology companies; healthcare providers such as doctors and nurses; hospitals; and health insurers. More respondents said biopharma companies had a “high” reputation compared with responses for insurers. But biopharma ranked last or tied with insurers for last on questions of whether each industry “puts patients over profits,” “makes a positive difference in the country,” and engages in “socially responsible behavior.” Source: The Harris Poll
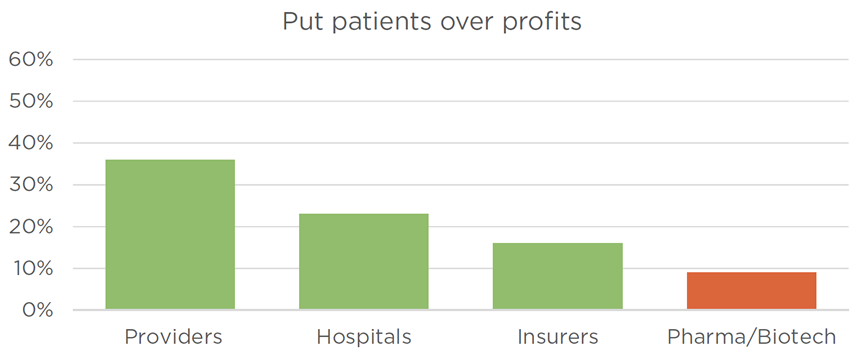
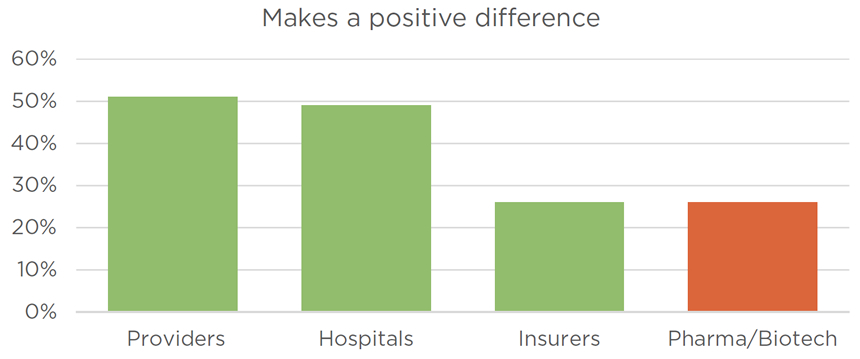
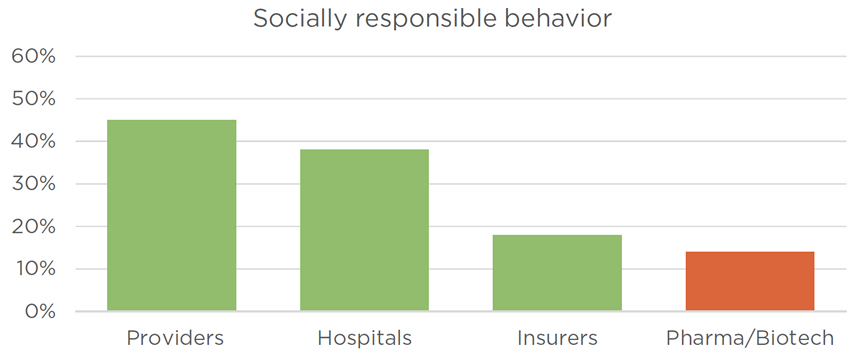
Figure: Cutting the check
While government spending on R&D for public health has leveled off in this decade, society’s investment in the life sciences remains significant. The U.S. is by far the global leader, with the NIH investing $303 billion over the past 10 years.
According to data from the Organization for Economic Cooperation and Development (OECD), government budget appropriations or outlays for R&D (GBAORD) for the U.K. totaled $26.5 billion, while Germany invested $13.9 billion and France $12.3 billion.
Asian healthcare-related R&D spending is quickly catching up to and surpassing that of continental Europe. Korea invested $14.1 billion, while Japan spent $13.7 billion and Taiwan $11.9 billion.
OECD data are not available for China. However, the Chinese government committed to invest $309 billion in science and technology between 2011 and 2015 as part of its 12th five-year plan, and consultancy ChinaBio LLC estimates the country dedicated over $200 billion to life sciences during the period. ChinaBio estimates the government’s 2016 investment in life sciences at more than $100 billion, and expects the 2017 figure to surpass $100 billion as well.
Below, OECD GBAORD figures for selected countries (right axis) are plotted against NIH actuals (left axis). GBAORD figures focus on the socio-economic objective of “health” and therefore may underrepresent spending on basic life science research. OECD figures adjusted to 2010 dollars and purchasing power parity (PPP). (A) Fiscal year ended Sept. 30; (B) 2016 GBAORD data not available; Sources: NIH; OECD
The path forward
Society will withdraw its support if there is a consensus that drug companies aren’t living up to their end of the deal. This is already starting to happen in the U.S., where some states have passed R&D transparency and anti-price gouging laws, some have introduced bills that include price caps, and efforts to impose price controls are gaining momentum. All of these measures are based on the notion that the public needs to be protected from predatory drug companies.
In Washington, heavily funded lobbying efforts to influence the political process only have produced a stalemate. Drug companies can often block laws, but have had limited success in advancing a positive agenda that is visible to the public.
Some of the industry’s top legislative priorities have stalled because they are widely perceived as efforts to improve the financial health of biopharma companies rather than help patients.
For example, the biopharma industry has failed to persuade Congress to either exempt drug patents from, or modify the standards for adjudicating approved patents under the inter partes review system. Abuses of IPR pose an “existential threat” to the biopharma industry, according to a letter by BIO President and CEO Jim Greenwood that BioCentury published June 1.
BIO’s concerns about the potential for IPR to scare off investment or unfairly invalidate valid patents may be overstated. Nonetheless, the industry’s inability to persuade Congress to move on a top-priority issue is telling.
“The fact is that too many people can’t afford the medicines that they need.”
Industry also lost battles to block right-to-try legislation in 37 state legislatures. The U.S. House of Representatives soon is likely to pass an RTT bill that the Senate passed in August. While the laws do not compel companies to provide access to drugs, they are part of an antiregulatory agenda that seeks to weaken, or in this case circumvent, FDA oversight. They likewise are an expression of public exasperation with difficulty accessing medicines.
That said, there have been some notable successes as governments seek ways to encourage drug companies to keep pursuing the search for treatments. One example is EMA’s adaptive licensing pathway, which seeks to enable swift approval of drugs for limited populations based on smaller than usual data sets followed by label expansions based on data including real-world evidence. Japan’s Sakigake accelerated approval pathway, coupled with reimbursement incentives for innovative drugs, is another (see “Japan’s Innovation Pothole”).
In Washington, the past year has seen political, industry and regulatory agreement on provisions in the 21st Century Cures Act and FDA user fee reauthorization legislation that could streamline and modernize medical product development.
Made possible by close collaboration with regulators and patients, these successes point to a winning formula: explicitly coupling policy proposals to benefits for individual patients and public health.
What won’t work is abdicating responsibility by pointing fingers and devising ever more exquisitely crafted attacks on payers, PBMs and health technology assessment agencies.
Neither are exhortations to “go boldly,” reminders that “time is precious,” or any other advertising slogans going to cut it.
To restore public confidence and justify society’s continued investment, biopharma companies must abide by -- and be seen abiding by -- the social contract.
First, the industry must make choices that put patients first.
Second, companies must publicly align management incentives with patient and public health, not just shareholder returns.
Third, and most obvious, drug companies must be seen improving access to medicines.
Sidebar: Japan’s innovation pothole
Bad choices
To fulfill the social contract, Back to School argues that drug companies must stop making choices that belie the industry’s claims that it puts patients first.
The drug industry has made its own bed of thorns through a host of CHOICES that violated the social contract. If it wants to restore its standing in society, the industry will have to make different choices. The alternative is accepting the consequences of the choices it has made.
The most difficult choice is between duty to patients and the fiduciary duty that CEOs and boards have to their shareholders. Back to School does not argue fiduciary duty is not real, or that it is not a legal reality. But prioritizing shareholders over patients ultimately is self-destructive and thus ruinous to the creation of long-term value for shareholders.
Almost no one, even inside the industry, believes that any nation has the financial ability to pay for the increasing number of breakthrough treatments under today’s pricing paradigm. This means companies are trapped between shareholder demands for continued extraordinary yield, primarily dividends and capital buybacks, and society’s inability -- and now unwillingness -- to pay the prices required to keep the yield engine humming.
Some pharma CEOs, however, continue to defend the status quo. In March, Pfizer Inc. Chairman and CEO Ian Read said in remarks at the National Press Club, “Overall, I think it is fair to say we are being responsible when it comes to the pricing of our medicines. We are producing great value for society and simultaneously taking large financial risk.”
As justification, he cited familiar tropes about the high cost of R&D, long timelines, low success rates and the small proportion of healthcare expenditures accounted for by drugs.
Read argued that, contrary to public perceptions, pharma is not a very profitable industry. “Our return on capital as an industry is around 11%,” a figure that he said is “not that healthy.”
BioCentury asked, but Pfizer did not answer, whether Read’s position has since changed.
Pricing drugs no longer can be an exercise where pharmas titrate to the highest level insurance companies and PBMs are willing to pay or a patient is willing to sacrifice.
The drive to prop up shareholder returns has led pharma companies to make choices that violate the social contract.
First among these choices is flattening the rate of spending on R&D.
The biggest pharmas consistently return more capital to yield-seeking investors than they spend on R&D. The combined value of dividends and buybacks has outpaced R&D investment over the past decade at $403 billion for the former to $389 billion for the latter (see “Follow the Money).
Back to School acknowledges this is not wrong in and of itself, as long as patients also are benefiting from the industry’s investment in innovation. But when drug companies flatten spending on R&D to maintain the payout to shareholders, management and boards have lost the right to say their top priority is innovation in the public interest.
Companies and their trade groups may continue to claim that investors won’t fund risky R&D without these extraordinary returns, but so far the data refute the claim: investors have plowed unprecedented sums into biotech over the past 10 years - more than $365 billion -- even as the drug pricing storm mounted.
In any case, the public has heard and rejected that argument.
Companies have even less defense for other profit-boosting choices.
Right at the top: Increasing the prices of drugs annually beyond any objective measure of increasing demand in the marketplace, or the rate of inflation generally, or within the healthcare system specifically.
While the prevalence of certain diseases may be increasing, it is impossible to argue that the industry’s unrelenting pace of price increases is driven by the laws of supply and demand. Nor is it possible, in the vast majority of cases, that the same drug has somehow increased in value.
Even less admirable: Accelerating the rate of price increases in the waning years of a drug’s patent life. This squeezes profits out of the system without providing value, leaves less room to pay for new innovations and results in higher prices for generics.
And one of the most cynical: Manipulating the regulatory system to increase a drug’s exclusivity when its patent life is at the end. REMS, for example, should be used as they were intended -- to protect patients -- not to deprive them of affordable alternatives after a company has enjoyed the monopoly permitted under law.
“We made a compromise to allow innovators to price at a premium to reward the substantial risk-taking in developing products,” Gottlieb told BioCentury. “We are willing to accept that trade-off as a society based on the expectation that the period of exclusivity is time-limited. When it is not, it undermines the whole premise of the contract we’ve entered into.”
The drug industry can’t absolve itself by disavowing the egregious actions of outliers such as Marathon Pharmaceuticals LLC and Turing Pharmaceuticals AG. These bad apples exacerbated but did not cause the destruction of industry’s reputation.
Even when companies are working on new paradigms for value, pricing and access, they can be seen to be engaging in self-serving behaviors. For instance, Novartis is a pathfinder in at least two innovative value-based pricing experiments that aim to increase access to Kymriah and heart-failure drug Entresto sacubitril/valsartan (see “Pathfinding for Access”).
At the same time, the pharma’s Gleevec imatinib provides one well-studied example of the pricing policies that have turned the public and physicians against drug companies. When Gleevec was launched in 2001, its ability to turn chronic myelogenous leukemia (CML) from a death sentence into a manageable chronic disease was hailed as a breakthrough.
In an extraordinary demonstration of enthusiasm, HHS Secretary Tommy Thompson held a press conference to announce FDA’s approval. There was only a little grumbling about the average worldwide price of $26,000 per year, and $30,000 in the U.S.
In a book about Gleevec’s development, former Novartis Chairman and CEO Daniel Vasella said the launch price was intended to allow the pharma to recoup its development costs in about two years.
By the time Gleevec patents expired in the U.S., the price was over $146,000 per year.
In a practice that is typical across the industry, and that clearly violates any commitment to patients, much of the price increase occurred in the three years prior to the U.S. patent expiration (see “Out with a Bang”).
The innovator price escalation also has had the perverse effect of making the generic versions more expensive. In August 2017, more than a year after generic imatinib launched in the U.S., the cheapest generic cost about $3,800 per month, which works out to $45,600 per year -- more than 50% higher than the branded drug’s launch price.
Meanwhile, Novartis continues to provide co-pay assistance that encourages patients to stick with the branded product, which costs more than twice as much as the generic, although there is no evidence that it is better than the generic.
This may reduce out of pocket expenses for patients while simultaneously preserving the company’s bottom line, but the tactic is objectionable in at least two ways.
First, it only shifts the financing burden to other stakeholders, in this case PBMs and plan sponsors. Society ultimately pays because the money that would have been saved by generics could have been applied to other urgent medical needs.
According to a June report from the Congressional Research Service, co-pay coupons overall are associated with both a decrease in generic substitution and with increases in the prices of branded drugs.
Second, propping up declining brands with subsidies invites attacks on other forms of patient assistance that Back to School argues are essential to assuring patients have access to important innovator drugs. These programs are necessary in the face of public and private payers’ use of co-pays and high deductibles to ration effective treatments while pinning the blame on drug manufacturers.
Dubious price escalation isn’t limited to cancer drugs.
According to the American Diabetes Association, the “cost of insulin nearly tripled between 2002 and 2013” and Americans are being forced to choose between rationing insulin and doing without other basic necessities.
“Rationing of insulin is a life-threatening practice and increases the risk of the horrific complications of diabetes including blindness, amputation, kidney failure, heart disease and stroke,” the ADA noted in November 2016.
Also last year, the U.S. Centers for Disease Control and Prevention reported nearly one in five Americans aged 45-64 with diabetes reduced or delayed medication to save money.
Insulin companies say they increase list prices to compensate for rebates captured by PBMs. For example, from 2001 to 2016, the list price of a vial of Novolog insulin aspart from Novo Nordisk A/S increased 353% in the U.S., while the net price increase - the money Novo retained -- was 36%, according to the pharma.
That’s slightly below the cumulative rate of inflation for the period, which was 38%. But diabetes patients who have plans that tie co-pays to list prices don’t care about that excuse, and Novo has since committed to capping price increases.
Back to School acknowledges that drug companies have no control over co-pays and co-insurance, which are dictated by insurers attempting to control their own costs. But that fact does not absolve drug companies of the impact of list price increases on the patients they are supposed to serve. Rather, it highlights the imperative for drug companies to work with other healthcare stakeholders on new value-based systems of pricing and reimbursement that ensure access to medicines that work (see “Rationing by Any Other Name”).
Bad choices about pricing aren’t the only problem: the public and politicians are incensed by many of the drug industry’s routine practices that they believe limit access and raise costs.
Individual companies continue to sully the entire industry by using legal loopholes to prolong monopolies, for instance, by abusing REMS and cutting deals to delay the introduction of generic drugs.
Even Gottlieb, hardly a card-carrying pharma-basher, has called out manufacturers of innovative drugs for stifling competition. Soon after he was sworn in, Gottlieb accused drug companies of misusing REMS and using other tactics to prevent generic and biosimilar manufacturers from obtaining drug samples, and prolonging negotiations with generic firms over the implementation of shared REMS systems to delay competition.
In July, Gottlieb told Congress that FDA is aware of more than 150 instances where a manufacturer used a REMS to block access to drug samples needed to develop a generic drug.
“Branded companies are using our rules that are intended to protect consumers, or meant to make the regulatory process more predictable, and taking advantage of these rules in order to deliberately forestall the entry of expected generic drug competition. In other words, they are ‘gaming’ our system,” he told Congress.
Gottlieb, a former venture partner at VC firm New Enterprise Associates and formerly a fellow at the American Enterprise Institute, said generic competition reduces cost and increases access.
Gottlieb told BioCentury that FDA will try to stop abuse of REMS through regulations. He added, however, that “the bigger phenomenon” is the use of contracts with “supply chain intermediaries” to restrict the ability of generic companies to obtain the drug substance they need to develop their low-cost alternatives.
These and other drug industry tactics put the business model for innovation at risk, according to Gottlieb.
“I support a market-based model for pricing innovation,” he said. “That model is predicated on a period of exclusivity granted to innovators that has a clear end.”
When drug companies game the system to hinder generic competition, he added, it “diminishes people’s willingness to support the market-based model of pharmaceutical innovation.”
In addition to routine misbehaviors by individual companies, the industry has collectively chosen take-no-prisoners positions on public policy issues that have helped destroy the reputation of the biopharma sector, and burned up much of its political capital.
The industry’s history of spectacular own-goals stretches over decades. They include opposing Nelson Mandela in a dishonorable effort to prevent South Africa from importing low-cost AIDS drugs, and a futile attempt to maintain perpetual monopolies on biologics in the U.S. by arguing that FDA violated the U.S. Constitution by even thinking about a biosimilar application.
More recently, industry blew off the opportunity to work with CMS to define a value-based purchasing system for Medicare Part B drugs. Instead of trying to improve a flawed plan, it defended an indefensible reimbursement system that rewards use of the most expensive medicines by pegging physician reimbursement for drugs to the average sales price (ASP).
The list goes on, including opposition to data transparency proposals in the EU and U.S., the Orphan Drugs Act and the legislation that created pediatric Priority Review vouchers (see “Own Goals”).
Sidebar: Rationing by any other name
Figure: Follow the money
Industry’s argument that high drug prices are required to help recoup R&D investments has never looked flimsier. From 2006 to 2016, sales at the 11 pure-play pharmas and biotechs in the Standard & Poor’s 500 index increased at a compound annual growth rate (CAGR) of 5.8%, reaching $219 billion last year. Much of this top-line growth has been derived from hefty annual price increases on existing drugs, and the introduction of new specialty products with eye-watering price tags.
Growth in SG&A has been held well below revenue growth at 2.6% CAGR, and SG&A as a percentage of revenue has dropped nearly 870 basis points to just 23.6% (top chart).
As companies have maintained sales growth while expanding margins, the added profit has not been recycled into greatly expanded R&D. Spending on R&D has grown at a 5.2% CAGR since 2006 to reach $47.4 billion last year. As a percentage of total sales, R&D has consistently hovered in the 20% range.
Shareholders are the primary beneficiaries of this dynamic. Dividends have increased at a 4.8% CAGR since 2006 -- roughly in line with sales and R&D growth -- hitting $22.6 billion last year. On the other hand, buybacks have accelerated robustly -- climbing at a 9.4% CAGR since 2006 to reach $26.8 billion in 2016 (bottom chart).
The combined value of dividends and buybacks has outpaced R&D investment over the past decade at $403 billion to $389 billion. Since 2011, the combined dollar amount of dividends and buybacks has been larger than R&D expenditures each year. Analysis includes acquired in-process R&D (IPR&D) expenses. Source: BCIQ: BioCentury Online Intelligence, SEC filings, company reports
Figure: Out with a bang
BioCentury analyzed six brands that achieved over $4 billion in worldwide peak sales. The average wholesale price (AWP) cumulatively increased an average of 166.1% over the 10 years prior to U.S. patent expiry. Within the final three years of market exclusivity, AWPs increases averaged 33.1%.
Gleevec imatinib mesylate from Novartis AG (NYSE:NVS; SIX:NOVN) led the group on both metrics, increasing 283.1% in aggregate over the 10 years before its patents expired and 45.2% within the last three years.
Two brands have seen routine price increases following genericization. The AWP for Lipitor atorvastatin from Pfizer Inc. (NYSE:PFE) has increased an average of 8.1% annually since 2011, translating to a 155.5% cumulative increase following patent expiry. Similarly, the once-daily formulation of Copaxone glatiramer acetate from Teva Pharmaceutical Industries Ltd. (NYSE:TEVA; Tel Aviv:TEVA) has increased 7.9% each year since patent expiry in 2015.
Abilify aripiprazole is marketed by Bristol-Myers Squibb Co. (NYSE:BMY) and Otsuka Pharmaceutical Co. Ltd.; Seroquel quetiapine by AstraZeneca plc (LSE:AZN; NYSE:AZN); and Plavix clopidogrel by Sanofi (Euronext:SAN; NYSE:SNY) and BMS.
AWPs are indexed to $1 per unit. AWPs are fixed at a 20% premium to wholesale acquisition cost (WAC); increases in AWP thus reflect list price increases by the manufacturer. Source: BCIQ: BioCentury Online Intelligence, Red Book, company reports
Table: Own goals
The pharmaceutical industry has a long history of alienating patients and potential allies by aggressively pursuing self-destructive public policies. For example, in the late 1990s, after three years, an unknown number of unnecessary deaths, countless ugly headlines and an outpouring of activism had darkened its reputation, the pharma industry admitted that South African imports of cheap AIDS drugs were legal under international trade law and dropped litigation that sought to stop it.
In a more recent example, industry negotiated with Congress to create an FDA biosimilars pathway only after it became clear that killing biosimilars outright was not possible. Industry’s opposition caused the U.S. to cede commercial and regulatory preeminence to Europe, and squandered an opportunity to foster competition that could have blunted the demand for price controls.
Even laws that are now celebrated by drug companies -- like the Orphan Drug Act -- were passed over the objections of pharma companies. “The pharma industry made the same mistake on the Orphan Drug Act that it did on any legislation introduced by Henry Waxman,” Abbey Meyers, the founder of the National Organization for Rare Disorders (NORD), told BioCentury. “If Waxman proposed it, the industry’s knee-jerk reaction was to oppose it, and believe me they pulled out all weapons to stop the ODA from being enacted.”
In many cases industry has reversed its position, embracing laws or regulations it tried to kill. According to Nancy Goodman, PhRMA lobbied against the Creating Hope Act. The bill has since led to biopharmaceutical companies trading more than $1 billion in Priority Review vouchers for rare pediatric diseases. Goodman, the founder of Kids v Cancer, conceived of and led efforts to enact the bill.
Industry continues to oppose policies that are sure to be implemented and unlikely to produce any harm, such as proposals by EMA and FDA to release de-identified patient-level data from clinical trials. The European Federation of Pharmaceutical Industries and Associations (EFPIA) and PhRMA want, and EMA has rejected, a gatekeeper to evaluate third-party researchers’ analysis plans before granting access to data. Seeking to circumvent EMA’s and FDA’s proposals, PhRMA and EFPIA have adopted principles on data sharing that give companies discretion to deny requests that would help competitors.
Selected examples of industry’s policy black eyes are below.
| Policy/law | Country/region | Key provisions | Industry position | Outcome |
| Biologics Price Competition and Innovation Act (BPCIA) | U.S. | Created pathways for both biosimilars and interchangeable biologics | BIO: There is no way to determine the active ingredients of a biologic or use analytic methods to determine similarity | Congress passed BPCIA in 2009, creating a pathway for biosimilars and interchangeable biologics. The first biosimilar wasn’t approved in the U.S. until 2015, nine years after biosimilars were introduced in Europe. |
| BIO: Referencing another company’s data -- even after patent expiry -- constitutes “use of others’ work without just compensation” | ||||
| PhRMA: Approving biosimilars would constitute unconstitutional property confiscation | ||||
| BIO, PhRMA, several biologics manufacturers: Interchangeability is scientifically impossible | ||||
| Clinical Trial Regulation No. 536/2014 | EU | Release de-identified patient-level data from clinical trials | EFPIA, PhRMA: Unvetted third-party analyses could cast doubt upon regulatory decisions | EMA policy on transparency went into effect in 2014 but implementation has been delayed until 2019 pending creation of IT systems; a similar proposal by FDA has stalled |
| Creating Hope Act | U.S. | Provides Priority Review vouchers for rare pediatric drugs | PhRMA: Lobbied against the bill in 2012, then strongly supported its reauthorization in 2016 | Enacted in 2012 as part of the FDA Modernization Act, and reauthorized in 2016 |
| HHS rule on data transparency | U.S. | Requires inclusion of summary results on ClinicalTrials.gov for all studies of products in Phase II and up, regardless of product approval status | BIO: Disclosure would not add value to the website and would divert resources from drug R&D | Proposed in 2014, implemented January 2017 |
| Medicare Part B value-based drug purchasing demonstration project | U.S. | Attempted to replace Part B reimbursement formula, which uses an add-on payment based on a percentage of average sales price (ASP), with a value-based payment system | PhRMA and BIO: The proposal would limit access, stifle innovation, and value-based pricing should be worked out in the private sector. The trade groups collaborated with physician groups to demand the proposal’s complete withdrawal. | Following intense bipartisan opposition, CMS withdrew a proposed rule that would have created the demonstration project |
| Medicines and Related Substances Control Amendment Act No. 90 | South Africa | Compulsory licenses on AIDS drugs | PhRMA: Lobbied White House to impose trade sanctions to force South Africa to abandon practices that could erode IP protection for drugs | PhRMA dropped the litigation after three years |
| PhRMA: Sued the South African government to prevent it from importing low-cost AIDS drugs | ||||
| Orphan Drug Act | U.S. | Creates incentives for development of drugs to treat rare diseases | Pharmaceutical Manufacturers Association (now PhRMA): Lobbied against the act behind closed doors because it didn’t believe producing drugs for rare conditions would be profitable, and it distrusted Henry Waxman (D-Calif.), who introduced the legislation | Enacted in 1983, followed by similar legislation in Japan and the EU |
Management incentives
Companies must publicly align management incentives with patient and public health, not just shareholder returns.
The litany of bad choices begs the question of what are good choices. One place to start is to create incentives for management and boards to choose public health over short-term shareholder yield.
The most visible place to accomplish that is in pricing, where some companies have at least started to revise the narrative.
Pricing drugs no longer can be an exercise where pharmas titrate to the highest level insurance companies and PBMs are willing to pay or a patient is willing to sacrifice.
Brent Saunders, chairman, president and CEO of Allergan plc, recognized this in a September 2016 blog post titled “Our Social Contract with Patients.”
“The health care industry has had a long-standing unwritten social contract with patients, physicians, policy makers and the public at large,” Saunders wrote. He said companies that have taken “aggressive or predatory price increases” have violated the contract, and made specific commitments for Allergan to honor that unwritten deal.
The element of his pledge that received the most attention was a cap on price increases in the U.S. to a single-digit percentage hike no more than once a year. Saunders said he expected the overall increases, net of rebates and discounts, would be in the low-to-mid single-digit percentages, “slightly above the current annual rate of inflation.”
Saunders added that Allergan would refrain from increasing the prices of drugs nearing patent expiration unless there was a corresponding increase in the company’s costs.
“While we have participated in this industry practice in the past, we will stop this practice going forward,” he said.
“More transparency on outcomes and cost information on what matters to patients and payers would really help.”
In January 2017, Allergan announced list price increases that averaged 6.7%. The company said the price increase for every product was below 10%, that it expects the net increase across its portfolio to be 2-3% after discounts and rebates, and that it would not take any additional price increases this year.
Several companies are getting on board with the idea that wanton price increases are undermining the industry’s reputation and must cease.
“The real reason we’re not liked, in my opinion, is because we as an industry have used price hikes to cover up the gaps in innovation,” Leonard Schleifer, CEO of Regeneron Pharmaceuticals Inc., said at the Forbes Healthcare Summit in New York in December 2016. “I hate us also when I see all this stuff.”
When Pfizer’s Read responded that drug costs as a percentage of healthcare spending have remained constant for the past two decades, Schleifer shot back: “You’re not entitled to a fraction of the GDP.”
Regeneron’s marketing partner Sanofi is one of at least six companies that have joined Allergan in vowing to cap list price increases.
In May, Sanofi published its principles on drug pricing and said its “guiding principle” would be to limit total annual list price increases to the rate of medical inflation using the U.S. National Health Expenditure growth rate as its benchmark. Sanofi said the growth rate this year is projected to be 5.4%.
Johnson & Johnson did not take up Allergan’s pledge, but did say it has been keeping annual list price increases in the U.S. in the single digits since 2012, with net price increases close to inflation levels.
Merck released similar data going back to 2010. And both pharmas committed to updating their price transparency reports annually.
To live up to the social contract, more companies will have to sign on, and all the companies will have to do a better job of keeping their commitments than the last time such pledges were made.
In 1990, when Vagelos pledged to keep Merck’s drug price increases at or below the rate of inflation, the rest of the industry responded to his implied challenge with deafening silence -- until the threat of price controls started to look real.
In March 1993, after the Clinton administration began advocating explicit drug price controls as part of its healthcare reform plan, PhRMA (then called the Pharmaceutical Manufacturers of America) asked the U.S. Department of Justice for an exemption from antitrust laws that would allow it to enforce a voluntary pledge by member companies to cap price increases at the level of inflation.
At the time, nine of the trade group’s members had made pledges similar to Merck’s; by year end another dozen companies had joined.
The antitrust waiver was denied, but a 2006 study by RAND Corp. researchers found that most companies honored the pledge, at least until the Clinton healthcare plan collapsed.
In 2004, Vagelos warned pharmaceutical companies that their pricing was putting their futures at risk. “High prices for drugs without profound medical value are turning people against the industry,” he said in remarks to the Wharton School at the University of Pennsylvania.
When companies are under strong pricing pressure, the only way to sustain and build the top line is to bring more new products to market, according to Jeremy Levin, chairman and CEO of Ovid Therapeutics Inc. He argues that biopharma executive compensation needs to be tied to the creation of breakthrough products that advance healthcare.
In a guest article in BioCentury in September 2016, he noted that senior management is often compensated based on EPS and total shareholder return. That creates incentives for actions that conflict with the social contract.
“We need to create compensation tools to reward successful pipeline investment, not just top-line growth and short-term stock appreciation,” he wrote.
Because executives inherit the investments of their predecessors, these tools would include both incentives and “clawbacks” to reward or penalize past decisions, he wrote.
Levin is a member of BIO’s executive committee. His experience includes serving as president and CEO of Teva Pharmaceutical Industries Ltd. and as SVP for strategy, alliances and transactions at Bristol-Myers Squibb Co.
Any executive compensation scheme should give CEOs freedom to maintain robust investments in R&D and encourage them to find other ways to maintain or even increase profit margins.
Beyond the obvious negative effects on pipelines, flattening R&D investment invites government intervention.
During the 2016 U.S. presidential campaign, Hillary Clinton proposed to “require pharmaceutical companies that benefit from federal support to invest a sufficient amount of their revenue in R&D.” Companies that failed to do so should be required to increase investment, or pay rebates to support basic research, she said.
U.S. politicians today are questioning the industry’s commitment to research, accusing it of free-riding on NIH-funded science and proposing standards for minimum corporate R&D investments.
This thinking leads straight to the sort of demands for transparency that harm innovators without benefiting patients at all, such as the demands to disclose product-level R&D expenses written into several U.S. state drug pricing bills.
No company should be compelled to reveal product-level R&D. But Back to School argues that drug company CEOs can use transparency to improve the efficiency of R&D, providing more headroom for margins as prices continue to be squeezed.
As Back to School discussed last year, many of the bottlenecks slowing development could be eased by the precompetitive sharing of data. The development of biomarkers is one place where this almost certainly is true.
Data transparency also would reduce the replication of futility as companies fail to learn from each other and expensively repeat mistakes or end up in dead ends that could have been avoided had information been shared.
J&J has already moved in this direction. It is providing third-party researchers with access to full clinical study reports and de-identified patient-level data from all trials conducted of drugs approved in the U.S. and Europe, trials of drugs that were terminated and trials that have been accepted for publication.
CEOs also should use transparency to mute accusations of secrecy, which feed society’s mistrust.
Supporting public release of FDA complete response letters would be a good place to start. CRLs typically include FDA’s explanation for why a marketing application can’t be approved and a description of what a sponsor would need to do to gain approval in the future.
Gottlieb told BioCentury in June that he favors redacting the letters and making them public. “There might be other products on the market or other products in development that are affected by the agency’s judgment,” he said.
Routine publication of CRLs and other documents would allow the scientific and medical communities to assess the reasonableness of the agency’s actions.
And the experience in Europe suggests that doing so would have no adverse effects. EMA routinely notifies the public about negative opinions and refusals of marketing authorization applications for human medicinal products. It publishes Q&As describing the grounds for a negative opinion, and publishes redacted versions of refusal assessment reports.
Access, access, access
Most importantly, drug companies must be seen to be improving access to medicines.
Voluntary commitments to cap price increases are a show of good faith, but they do not solve access problems.
Back to School can’t provide a definitive list of concrete actions that will result in more access, because it’s a shared problem that will require collaboration among several industries and government. But the starting points for the drug industry are clear.
Improving access begins with reducing prices of some drugs, eschewing premium pricing for new drugs that provide only incremental improvements over standard of care, and eliminating price increases that are not justified by improvements in value to patients and health systems.
It includes refraining from abusing IP protections or exploiting loopholes to prevent cheaper generics and biosimilars from coming to market at the appropriate time.
Crucially, the winning formula includes working with other healthcare sectors to help solve systemic access problems -- even those that biopharma companies have not created -- including facilitating a shift to a transparent reimbursement system that fully rewards interventions that improve outcomes.
As Back to School has been arguing since 2013, the prices of newly launched drugs will have to be supported by objective assessments of value that justify reimbursement and thus ensure access.
“Access to medicines is only available when you truly demonstrate benefit. More holistic assessment will inform payers and patients, who increasingly contribute more, to care about the value that’s being delivered to them,” John Glasspool, an advisor to the New Drug Development ParadIGmS (NEWDIGS) program, told BioCentury.
NEWDIGS is housed within the Massachusetts Institute of Technology’s Center for Biomedical Innovation (CBI). The group laid the groundwork for EMA’s adaptive licensing pathway. Its current projects include work to design and pilot financing and reimbursement models for curative therapies, and to demonstrate how data obtained outside of randomized clinical trials could be used to develop meaningful outcomes measures and support regulatory and access decisions. Its collaborators include academics, regulators, drug companies, patients and payers.
“Access to medicines is only available when you truly demonstrate benefit.”
Glasspool was previously EVP and head of corporate strategy and customer operations at Baxalta Inc., which was acquired by Shire plc.
“There is a sense of urgency, mixed with frank awe” for the quality of innovation payers, providers and patients are seeing, noted Steven Pearson, founder and president of the Institute for Clinical and Economic Review (ICER). “Cures for hepatitis C and new CAR T drugs are the kind of innovation everyone welcomes. Yet they are worried because they don’t see how paying for these fits in with the other things they need to pay for.”
ICER is a non-profit organization that evaluates evidence on the value of medical interventions. It publishes suggested “value-based price benchmarks” that payers use in negotiating prices of drugs and devices.
“Drug companies have a responsibility to make defining value a collaborative process,” Pearson told BioCentury. “That doesn’t mean they need to accept someone else’s definition of value, but they do have to engage with other stakeholders.”
Pearson says there are signs that drug companies recognize the need for value-based pricing, and predicted they will find solutions that don’t involve price controls. However, he warned, “If pharmaceutical manufacturers continue to have this much power over prices and society feels it is being abused, the system will be changed and European models imposed.”
While Saunders did not explain how, he explicitly committed Allergan to pricing “products in a way that is commensurate with, or lower than, the value they create by mitigating or avoiding the need for other treatment modalities or providing better quality of life to those patients without other treatment options.”
Sanofi said it would consult with “external stakeholders” and would base its launch prices on a “holistic” assessment of value, including clinical outcomes, economic value and social value. It defined the latter as improvements in quality of life and productivity.
The pharma said its pricing process for new medicines also would take into account similar treatment options that are or will soon be available; affordability for patients, payers and healthcare systems; and factors such as the need to fulfill regulatory commitments, conduct additional trials and develop sophisticated patient support tools.
While such approaches may dovetail with the health technology assessment (HTA) model that drives many single-payer systems, the drug industry is falling behind in the U.S., which is the major reservoir of global profits, as the healthcare payment system moves to replace the fee-for-service model.
Former CMS and FDA chief Mark McClellan, now director of the Robert J. Margolis Center for Health Policy at Duke University, told BioCentury drug companies aren’t doing nearly enough to move to value-based pricing, and said there is not enough transparency.
The American healthcare system is “going to episode payment models, new primary care payment models, and full capitation population risk models,” McClellan noted. “Drug and biologics manufacturers are not closely aligned with providers that are trying to make progress in that direction.”
PBMs and private payers in the U.S. including Aetna Inc., Cigna Corp., Prime Therapeutics LLC, and Harvard Pilgrim Health Care Inc. have been experimenting with outcomes-based contracting.
Express Scripts Holding Co. and CVS Health Corp. are testing indication-based payment models for drugs approved to treat more than one indication.
Harvard Pilgrim has tried to engage drug sponsors in a pilot of episode-based payment that would reimburse a flat amount for an episode of care, regardless of the amount of drug used. CMO Michael Sherman said he hasn’t gotten any uptake on that model. However, he added manufacturers are showing “more interest in the outcomes-based approach.”
Payers and PBMs have told BioCentury that biopharma companies could be subject to fewer restrictions and see a net increase in use of their drugs -- meaning more access for patients - if they joined in testing these new models.
Sadly, the number of biopharma companies that have disclosed their participation in value-based pricing deals remains insignificant. And last year, when the industry had a chance to test these models on a large scale within the Medicare Part B drug benefit, it not only balked, it went on the attack to kill the proposal.
McClellan acknowledged there are obstacles to value-based pricing in the U.S. that could be alleviated through legislation and regulation, but he argued these have been exaggerated. “There are ways to manage many of those perceived impediments,” he said.
McClellan served as CMS administrator and FDA commissioner in the George W. Bush administration, and currently serves on the board of J&J.
Novartis appears to have found a way around obstacles to value-based pricing for Kymriah via an outcomes-based deal with CMS. In its approved indication for children and young adults with acute lymphoblastic leukemia (ALL), the pharma will receive payment only for patients who respond to Kymriah by the end of the first month.
The arrangement also includes indication-based pricing, under which the terms of payment for future indications “would be reviewed for the most relevant outcomes-based approach.”
McClellan also told BioCentury reducing systemwide costs will require “better evidence about what we are getting for what we are spending. More transparency on outcomes and cost information on what matters to patients and payers would really help.”
To that end, drug companies should work with patients to produce data on outcomes -- including data that aren’t typically included on FDA labels -- that help them optimize their care. They also should collaborate with payers to generate information that would allow patients to assess the cost and performance of alternatives.
Back to School reiterates that a lack of transparency on prices invites suspicion and leads to false assumptions about the relationship between drug prices and company cost of goods and investment in R&D. But it is the industry’s own arguments that have led to this predicament. The constant drumbeat that high drug prices are needed to recoup R&D investments has fueled demands to disclose detailed costs associated with the development and manufacturing of specific drugs.
An early example came from the U.K. in 2014, when NICE tried to demand R&D costs to negotiate the price of Orphan drug Soliris eculizumab from Alexion Pharmaceuticals Inc. The agency said it was unable to make a recommendation on the drug because the appraisal committee could not justify its cost “in light of the manufacturing, research and development costs of a medicinal product for the treatment of a very rare condition.”
NICE did not succeed in extracting the R&D numbers from Alexion, and Soliris was eventually recommended with restrictions, but the idea that R&D cost could be used to determine value did not die there.
In the U.S., pharmaceutical cost transparency laws making their way through state legislatures are seeking to pave the way for drug price controls by exposing profit margins on high-priced drugs.
Other aspects of access are not within drug company control, and other healthcare players must be called out for practices that let patients and society down.
For example, if transparency is demanded of drug companies, what about the unfathomable prices charged by hospitals?
The PBM situation is particularly perverse as its business model is built on extracting rebates that do not necessarily contribute to lower payer or patient costs. In fact, the PBM model encourages drug companies to jack up list prices in anticipation of the demand for rebates.
This is one instance where drug industry management and boards owe it to their shareholders to hold a spotlight on profiteering.
However, a war on PBMs will not help patients unless both sides stop protecting their short-term P&Ls at all costs and begin to collaborate on solutions that address the social contract.
“I don’t believe that pointing fingers, blaming one industry, is at all constructive,” Ovid’s Levin told BioCentury. “For a long time several components of the system have been quite comfortable having the pharmaceutical and biotech industry take the blame for price rises. We all -- PBMs, distributors, insurers and pharmaceutical companies -- have a common responsibility to ensuring we bring affordable access to great medicines,” he said.
Novo has explicitly committed to collaborating with payers, PBMs, insurers, employers and patient organizations to simplify and improve the complex pricing system in a position paper about pricing and affordability posted on its website.
“The system we currently have is very complicated -- rebates, discounts, administrative fees, co-pays and deductibles all play a role in what our customers, and people with diabetes, pay. We need to work with all involved to simplify and transform the system,” the pharma wrote.
McClellan noted the system needs to change, because there is no reason to think prices would come down under the current paradigm without the negotiating leverage of PBMs.
“If you don’t like this non-transparent system for having very large purchasers negotiate to get prices down, what’s the alternative?” McClellan said. “One is to switch to a system based on value and the results patients are getting as an alternative to price negotiation.”
Consensus on a product’s value is critical for establishing a price, but it is not sufficient. For patients, value-based pricing still will be indistinguishable from extortion when a drug’s high price isn’t accompanied by guarantees about access.
In his pledge, Saunders committed Allergan to working with government payers, regulators, private insurers, and PBMs to make the company’s products accessible.
Saunders also vowed to expand Allergan’s patient assistance programs, to engage in responsible education about its products and to maintain quality and ensure availability of the pharma’s products. Other pharmas have made similar pledges.
Patient assistance programs that provide free drugs to patients who lack insurance coverage, or pick up some or all of the costs for deductibles and co-pays for eligible patients, are among the most powerful tools for drug companies to improve access.
However, according to Marc Boutin, they need to be more consistent, stable and easier to access.
Boutin is CEO of the National Health Council (NHC), an umbrella group representing patients with chronic diseases and disabilities. Its members include patient advocacy organizations, as well as biopharma companies and payers.
Drug assistance programs “are an incredible challenge for patients,” Boutin told BioCentury. “Programs differ from company to company, product to product. The rules on how they operate can change on an annual or quarterly basis. You need to know how to find them, which is difficult for the ordinary patient.”
He added: “At the same time, millions of American patients get access through these programs.”
A June report from the Congressional Research Service noted that according to tax records, total giving by patient assistance programs run by 10 large biopharma companies rose from $376 million in 2001 to $6.1 billion in 2014. Independent patient assistance charities increased their giving over the same period from $2 million to $868 million.
There is a clear need for more creativity around means-tested access. The bottom line is that no patient should be denied treatment because they can’t pay, and no one should be forced to sell their home or bankrupt their family to qualify for assistance.
The “Help at Hand” program from Takeda Pharmaceutical Co. Ltd.’s U.S. business provides an example to follow. Help at Hand provides 11 marketed drugs at no or reduced cost to Americans with incomes up to 400% of the Federal Poverty Level. For a family of four, the cutoff is $100,000 in annual income.
The application is simple and easy to find. It asks for insurance information, and for documentation of annual income. It does not ask about savings or other assets.
As of April 2016, Help at Hand had provided more than 1 million prescriptions to over 200,000 patients in the U.S. The company operates a separate assistance program for oncology products.
Takeda includes contact information for its assistance program on drug ads.
Just as it is unacceptable to deny access to life-saving drugs to low-income patients in wealthy countries, the social contract means it is not acceptable for patients in low-income countries to suffer and die because they cannot afford drugs.
Some companies also have implemented generous programs for ensuring access to medicines in poor countries, but drug manufacturers can’t solve problems of global access on their own. To fulfill the contract, more drug companies must do more to collaborate with governments, NGOs and industry in developing countries.
Companies also must improve access to unapproved drugs.
There may be legitimate reasons, including supply constraints, for restricting or denying pre-approval access, but manufacturers of breakthrough products have an obligation to attempt to overcome those constraints. When they can’t be overcome, companies must provide timely explanations to patients.
Failures to do so have reinforced public perceptions of biopharmaceutical companies as heartless profit-seekers and provided ammunition to ideologues with an antiregulatory agenda.
The right-to-try battle in the U.S. is based on the false premise that FDA is the principal roadblock to patients accessing unapproved therapies. In fact, access is almost entirely determined by companies that own the drugs.
Opponents and skeptics believe right-to-try legislation would harm patients by exposing them to untested therapies and delaying the collection of data that are essential for approvals. Transparent compassionate access policies, and large-scale expanded access programs, are the antidotes.
J&J provides one model. The pharma implemented a comprehensive pre-approval access policy that includes an independent advisory board that evaluates requests for compassionate use.
Action items
Even if the drug industry individually and collectively reforms itself, Back to School acknowledges it will take time to repair biopharma’s reputation.
The current reality is that the drug industry gets little or no recognition even when it scrambles to develop medical countermeasures to urgent public health threats.
One example is the industry’s extraordinary response to the Ebola epidemic. Several biopharma companies with unique capabilities put high-priority R&D on hold, diverted scientists from product development, collaborated transparently with governments and NGOs, and developed prototype vaccines in record time.
If something as stark as working to stop an epidemic in its tracks doesn’t capture the public’s imagination, it is unlikely that any changes in biopharma’s more routine business will spark a quick turnaround in its reputation.
As noted at the beginning of this essay, the marriage between the drug industry and its customers has been unwinding for decades. It will take a long time to restore that trust.
Rehabilitation will take actions, not words in press releases. Beyond the strategic approaches described above, like using transparency to mute accusations of secrecy, Back to School advocates this starter list of five action items for drug companies:
Actively and visibly demonstrate industry’s devotion to collaborating on value-based metrics for pricing and reimbursement.
Some work has begun here, such as Novartis’ deals with payers for Entresto. The pharma shares the risk by paying rebates based on whether hospitalizations exceed or drop below an undisclosed threshold. Hospitalization is a good metric for plans or payers that provide both pharmacy and medical benefits, because it hits their bottom line and is a proxy for health outcomes.
However, if a value-based system is to replace the current volume-based or fee-for-service approach -- as Back to School argues it must -- then companies will need to work with patients and payers on a much bigger scale to devise metrics for all diseases, or at least those responsible for the lion’s share of drug spend.
Support independent efforts to assess value.
Given that the public does not trust drug developers or insurers, third parties logically will need to serve as credible arbiters of value. In Europe and other parts of the world, health technology assessment bodies fill this role. In the U.S., ICER is hoping to do so.
Regeneron’s collaboration with ICER on its assessment of Dupixent dupilumab for atopic dermatitis is a step in the right direction. The biotech provided data to ICER, and in return received early access to the report’s conclusions for use in discussions with payers, according to Pearson.
Regeneron said ICER’s report factored into Dupixent’s $37,000 list price and average net price in the low $30,000s -- well below the most commonly cited comparators but still not “cheap” by consumer standards.
Express Scripts praised Regeneron and partner Sanofi’s approach to the launch as “responsible” and included Dupixent on its National Preferred Formulary.
The entire healthcare system, including drug companies, must do a far better job of generating data that can be used to make such assessments.
Biopharma companies have an “obligation to generate the best information about value that they can produce within the constraints FDA and the competitive landscape give them,” Pearson told BioCentury.
Drug developers can support this work by developing data on outcomes, including quality of life, that are important to patients and payers. Partnerships with healthcare providers whose business models now require them to demonstrate improvements in the quality of care they deliver are an appropriate vehicle for generating these data.
Integrated health systems and accountable care organizations (ACOs) top the list, along with Medicare Advantage providers, hospitals and large physician groups.
Collaborate with patient groups to create and stress-test access programs.
Access programs developed by companies in a vacuum are not sufficient; patient perspectives are essential for designing access programs and assessing their effectiveness.
Rare diseases provide the most abundant examples of effective approaches, in which most if not all companies work directly with patient groups, and even individual patients, to ensure access to drugs.
Manufacturers also provide funding to assistance programs run by patient groups, like the National Organization for Rare Disorders’ RareCare program. NORD uses the money to help patients with rare conditions obtain medicines; pay insurance premiums and co-pays; and obtain assistance with diagnostic testing, travel for clinical trials or consultation with disease specialists. NORD does the screening of patients for eligibility and disburses the money.
These models could be extended to more disease areas via collaboration with other patient advocacy and disease foundations.
Call détente with PBMs, insurers, governments, and providers that are willing to collaborate on patient access, while publicly calling out stakeholders that are unwilling to share responsibility for solutions.
Express Scripts, for example, has been willing to engage with drug manufacturers to design an indication-based pricing pilot in cancer. In 2015, the PBM told BioCentury it planned to work with manufacturers to discuss what drugs and indications are best suited to the pilot and to negotiate how reimbursement rates would be set for each indication.
As of December 2016, the PBM said the pilot included undisclosed drugs for four malignancies: multiple myeloma (MM), non-small cell lung cancer (NSCLC), prostate cancer and renal cell carcinoma (RCC). Express Scripts did not provide an update in time for publication.
Express Scripts also spearheaded a partnership with pharmas and pharmacies to provide discounted medicines to patients who are uninsured or have high out-of-pocket costs for branded drugs. The program provides an average discount of 34% on more than 40 branded drugs for common illnesses including diabetes, asthma, heart disease, depression, gastrointestinal disorders and gout.
Participating pharmaceutical companies include Boehringer Ingelheim GmbH, Eli Lilly and Co., H. Lundbeck A/S, Novo Nordisk, Sanofi, Takeda and Teva.
Finally, continue to create breakthrough medicines with patients as partners.
Restoring the trajectory of R&D budgets is a starting point. But to communicate its commitment to the social contract, the drug industry should do more to embed patients in the process beyond asking them to take risks in clinical trials.
Except for Orphan drugs, biopharma is one of the few successful modern industries that fails to systematically and rapidly incorporate feedback from customers to improve the way its products are delivered, developed and distributed.
As BioCentury has pointed out in previous Back to School essays, investing in patient preference research and including patient perspectives all along the drug life cycle would result in better products with more useful data, while eliminating investment in drugs patients do not want and will not use.
Executing on this beginning list of action items -- and identifying new ones -- would start to repair the biopharma industry’s relationship with society. And if more companies partner with patients and payers to prioritize clinical needs, define and demonstrate value, and agree on plans that ensure access to medicines, then these constituents will have a stake in protecting the industry that produces them.
At the end of the day, macroeconomics matter. In the absence of a transformational change in the drug industry’s cost structure, society’s limited ability to pay for drugs means that extraordinary profits will be forced down to merely above average. It’s not a matter of whether this happens, but when.
The bottom line is that continuing to prioritize for short-term shareholder interests will just hasten the day when societal mandates undermine the drug industry’s ability to generate sustainable returns for its investors.
Investors who demand outrageous returns will end up looking elsewhere, and there’s nothing the drug industry can do about that.
If the drug industry lives up to the social contract, then in return, society will find a way to reward true innovations. That’s the deal.
The 25th Back to School Commentary is a collaborative work co-written this year by Washington Editor Steve Usdin, BioCentury Editor Susan Schaeffer and President & CEO David Flores. New reporting was contributed by Steve Usdin and Senior Editor Erin McCallister. Data were developed by Amran Gowani and Meredith Durkin Wolfe, Senior Editor and Associate Editor of Research & Analytics, respectively.
Companies and Institutions Mentioned
Aetna Inc. (NYSE:AET), Hartford, Conn.
Alexion Pharmaceuticals Inc. (NASDAQ:ALXN), New Haven, Conn.
Allergan plc (NYSE:AGN), Dublin, Ireland
American Diabetes Association (ADA), Arlington, Va.
American Enterprise Institute, Washington, D.C.
Biotechnology Innovation Organization (BIO), Washington, D.C.
Boehringer Ingelheim GmbH, Ingelheim, Germany
Bristol-Myers Squibb Co. (NYSE:BMY), New York, N.Y.
Cigna Corp. (NYSE:CI), Bloomfield, Conn.
Congressional Research Service, Washington, D.C.
CVS Health Corp. (NYSE:CVS), Woonsocket, R.I.
Duke University, Durham, N.C.
Eli Lilly and Co. (NYSE:LLY), Indianapolis, Ind.
European Medicines Agency (EMA), London, U.K.
Express Scripts Holding Co. (NASDAQ:ESRX), St. Louis, Mo.
Harris Poll, Rochester, N.Y.
Harvard Pilgrim Health Care Inc., Boston, Mass.
H. Lundbeck A/S (CSE:LUN), Copenhagen, Denmark
Institute for Clinical and Economic Review (ICER), Boston, Mass.
Japan Pharmaceutical Manufacturers Association (JPMA), Tokyo, Japan
Johnson & Johnson (NYSE:JNJ), New Brunswick, N.J.
Kaiser Family Foundation, Menlo Park. Calif.
Marathon Pharmaceuticals LLC, Northbrook, Ill.
Massachusetts Institute of Technology, Cambridge, Mass.
Merck & Co. Inc. (NYSE:MRK), Kenilworth, N.J.
Ministry of Health, Labour and Welfare (MHLW), Tokyo, Japan
National Community Pharmacists Association (NCPA), Alexandria, Va.
National Health Council (NHC), Washington, D.C.
National Institute for Health and Care Excellence (NICE), London, U.K.
National Institutes of Health (NIH), Bethesda, Md.
National Organization for Rare Disorders (NORD), Danbury, Conn.
Novartis AG (NYSE:NVS; SIX:NOVN), Basel, Switzerland
Novo Nordisk A/S (CSE:NOVOB; NYSE:NVO), Bagsvaerd, Denmark
Ono Pharmaceutical Co. Ltd. (Tokyo:4528), Osaka, Japa
Ovid Therapeutics Inc. (NASDAQ:OVID), New York, N.Y.
Pfizer Inc. (NYSE:PFE), New York, N.Y.
Pharmaceutical Research and Manufacturers of America (PhRMA), Washington, D.C.
Prime Therapeutics LLC, Eagan, Minn.
RAND Corp., Santa Monica, Ca.
Regeneron Pharmaceuticals Inc. (NASDAQ:REGN), Tarrytown, N.Y.
Sanofi (Euronext:SAN; NYSE:SNY), Paris, France
Shire plc (LSE:SHP; NASDAQ:SHPG), Dublin, Ireland
Takeda Pharmaceutical Co. Ltd. (Tokyo:4502), Osaka, Japan
Teva Pharmaceutical Industries Ltd. (NYSE:TEVA; Tel Aviv:TEVA), Petah Tikva, Israel
Turing Pharmaceuticals AG, Baar, Switzerland
U.S. Centers for Disease Control and Prevention (CDC), Atlanta, Ga.
U.S. Centers for Medicare & Medicaid Services (CMS), Baltimore, Md.
U.S. Department of Health and Human Services (HHS), Washington, D.C.
U.S. Department of Justice (DoJ), Washington, D.C.
U.S. Food and Drug Administration (FDA), Silver Spring, Md.
The Wharton School at the University of Pennsylvania, Philadelphia, Pa.
References
Situation
Harris Poll. “Study of reputation equity and risk across the health care sector.” (2017)
Kaiser Family Foundation. “Kaiser health tracking poll: September 2016.” (2016)
Path forward
Greenwood, J. “Letter to BioCentury: BIO on IPR.” BioCentury (2015)
McCallister, E. “Access and innovation in Japan.” BioCentury (2017)
Usdin, S. “Beyond right to try.” BioCentury (2017)
Usdin, S. “Breaching the wall.” BioCentury (2017)
Bad choices
Centers for Disease Control and Prevention. “Morbidity and mortality weekly report.” (2017)
Chen, C. and Kesselheim, A. “Journey of generic imatinib: A case study in oncology drug pricing.” Journal of Oncology Practice (2017)
Gorkin, L. and Kantarjian, H. “Targeted therapy: Generic imatinib -- impact on frontline and salvage therapy for CML.” Nature Reviews Clinical Oncology (2016)
Schaeffer, S., et al. “Editors’ commentary: Save the baby.” BioCentury (2016)
Unsigned commentary. “Goodwill hunting.” BioCentury (2015)
Management incentives
Fisher Ellison, S. and Wolfram, C. “Coordinating on lower prices: pharmaceutical pricing under political pressure.” RAND Journal of Economics (2006)
Levin, J. “A call to change incentives.” BioCentury (2016)
Usdin, S. “Health & wealth.” BioCentury (2017)
Usdin, S. “Starting the conversation.” BioCentury (2017)
The Wharton School at the University of Pennsylvania. “Roy Vagelos talks about leadership and the need for new drug pricing policies.” Knowledge@Wharton (2004)
Access, access, access
Congressional Research Service. “Prescription drug discount coupons and patient assistance programs (PAPs).” (2017)
Hansen, S. “Slippery slope.” BioCentury (2014)
McCallister, E. “Refining moment.” BioCentury (2016)
Unsigned commentary. “Back to School: Paying the piper.” BioCentury (2014)
Unsigned commentary. “Don’t go there.” BioCentury (2014)
Unsigned commentary. “Back to School: Facing reality.” BioCentury (2013)
Usdin, S. “Is that clear?” BioCentury (2016)
Action items
Hansen, S. “Wac-a-mol.” BioCentury (2017)
Longman, R. “Contracting 2.0” BioCentury (2017)
McCallister, E. “Indications of value.” BioCentury (2015)
McCallister, E. “Paying in heart failure.” BioCentury (2015)