Product Development
There are opportunities to eradicate opioid misuse: Investors and innovators should seize them
Guest Commentary: NIDA Director Nora Volkow with a call to biotechs to help solve the exploding opioid crisis
Eradicating opioid misuse and addiction will take an integrated, holistic approach involving prevention, treatment, and recovery. Crucially, it will also take the help of investors and innovators in biotechnology who understand the challenges and opportunities that exist in this space.
More than 70,000 people died in the U.S. from drug overdoses in 2017, and the numbers continue to rise. Most of the deaths involve opioids, but this epidemic is rapidly evolving. The drugs implicated in the overdoses are changing, as are the demographics of those affected and the most affected geographic locations.
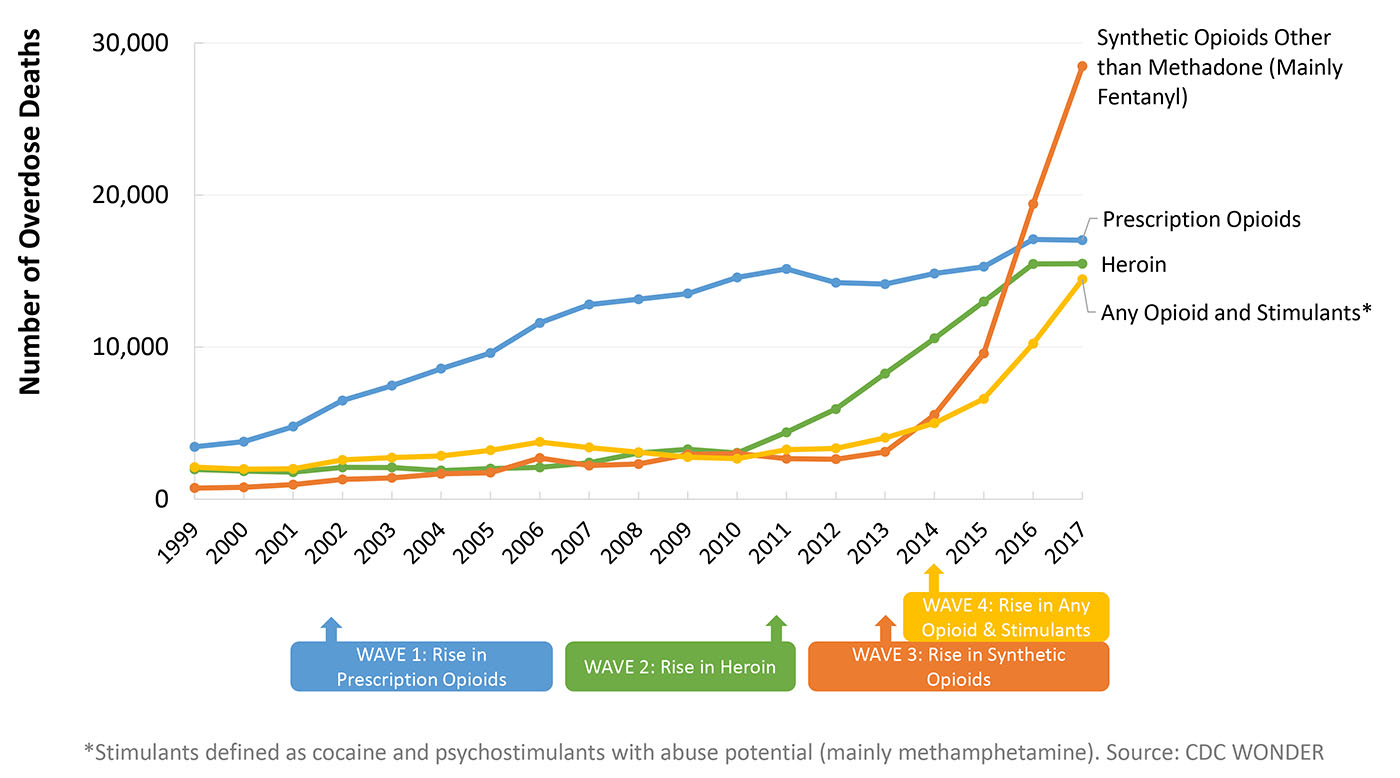
The epidemic began with overprescription, diversion and misuse of prescription opioid pain relievers (first wave). A rise in heroin consumption and overdoses followed (second wave), after which there was an emergence of overdoses from exposure to highly potent synthetic opioids such as fentanyl and its analogs (third wave). Alarmingly a fourth wave is appearing, involving overdose fatalities associated with opioids in combination with cocaine and methamphetamine (see Figure: “Waves of Opioid-Involved Overdose Deaths”).
A healthcare system that for decades avoided addressing substance use disorders must become more integrated and responsive.
Ending this epidemic of overdose deaths and reducing the number of people suffering from opioid use disorder (OUD) have become top priorities for federal agencies, as well as state and local government.
With greater recognition that addiction is a chronic and treatable disease and not a moral failing, a healthcare system that for decades avoided addressing substance use disorders must become more integrated and responsive.
And the recognition that addiction is a chronic disease of the brain frames the opportunity for developing effective medications and other therapies to treat it, as well as expanding access to treatments that are currently available.
Figure: Waves of opioid-involved opioid deaths
Four waves characterize the increase in deaths due to opioids. Wave 1, from 1990 to 2004, involved principally prescription analgesics; wave 2, from 2005 to 2010, involved a rise in heroin deaths; wave 3, from 2011 to 2016, involved a sharp rise in deaths from fentanyl , sometimes contaminating heroin or other drugs; wave 4, starting in 2014, involves a rise in deaths from cocaine or methamphetamine mixed with an opioid.
Wanted: New formulations, medications, modulators
Medications for the treatment of OUD (MOUD) are the standard of care; they significantly improve clinical outcomes, including reducing overdose risk as much as 70%.
Yet despite the strong evidence for their effectiveness, most people with opioid addiction are not treated with MOUD, and even when treated they are not provided with the sustained care necessary to remain in treatment. As a result, on average, 50% of those given MOUD will discontinue treatment with six months of initiation, putting them at risk not only for overdose death but also for other health consequences.
Those consequences can include infection with HIV, hepatitis C, and other diseases transmissible through sharing of drug-injection equipment or high-risk sexual behavior associated with drug use.
A recent review of the scientific evidence by the National Academies of Science, Engineering, and Medicine (NASEM) underscored that withholding FDA-approved MOUD is unethical, putting further pressure on addiction treatment providers and the rest of healthcare to follow evidence-based standards.
The same pressure is being put on a justice system that historically has denied effective medication treatment for OUD and put prisoners at high risk of relapse and overdose death upon community release.
The current standard of care for OUD utilizes FDA-approved formulations of methadone, a full μ opioid agonist; buprenorphine, a partial agonist; or naltrexone, an antagonist (see Table: “Medications for Opioid Use Disorder”).
Table: Medications for opioid use disorder
| Medication | Description | Pharmacology | FDA-approved formulations and dosing |
| Methadone | Used for more than four decades | Full μ opioid receptor (MOR; OPRM1) agonist | Daily oral dosing, at doses typically 80 to 160 mg |
| Dispensing mostly limited to licensed opioid treatment programs (OTPs) or “methadone clinics” | |||
| Reduces cravings and withdrawal symptoms | |||
| Buprenorphine | Approved to treat OUD in 2002 | Partial MOR agonist, κ opioid receptor (KOR; OPRK1) antagonist, opioid related nociceptin receptor 1 (OPRL1; NOPR) agonist | Immediate-release (IR) formulation: most frequently prescribed as a sublingual film that contains naloxone, which induces withdrawal when drug is injected; generally taken 3-4 times/week at daily doses typically between 16 and 24 mg |
| Dispensing by physicians or nurses who have a Drug Abuse Treatment Act (DATA)-2000 waiver | |||
| Reduces cravings and withdrawal symptoms | Extended-release (ER) formulations: a subdermal implant that delivers the equivalent of 8 mg of buprenorphine was approved in 2016; a once-monthly depot injection was approved in 2017, and additional once-monthly and once-weekly formulations are being reviewed for FDA approval | ||
| Extended-release (ER) formulations will facilitate compliance and OUD management, including for patients living in rural areas | |||
| Naltrexone | Approved for OUD in 1984 but had poor compliance until extended release ER formulation was approved for OUD in 2010 | MOR antagonist, KOR antagonist | IR formulation: 50 mg once daily |
| Does not require a license or waiver to prescribe | ER formulation: 380 mg delivered intramuscularly every four weeks | ||
| Interferes with the binding of opioid drugs, thus inhibiting their effects, including reward and analgesia | |||
| Patients need to be detoxified before initiating naltrexone to avert withdrawal, which can be challenging and not all patients succeed | |||
| Lofexidine | Approved in 2018 as the first FDA medication to treat opioid withdrawal, although it has been used to treat opioid withdrawal in the U.K. since the 1990s | α-adrenergic receptor agonist | Three 0.18 mg tablets taken orally 4 times daily at 5- to 6-hour intervals |
| Dispensing is by physicians | |||
| Naloxone | First approved for overdoses in 1971 | MOR antagonist | Autoinjection: 2 mg/0.4 mL naloxone hydrochloride solution in a pre-filled autoinjector for intramuscular or subcutaneous injection |
| Autoinjector was approved in 2014 | |||
| Intranasal spray was approved in 2015 | Nasal spray: 4 mg of naloxone hydrochloride in 0.1 mL for intranasal dosing | ||
| Increasing access to naloxone is a major component to reverse the overdose epidemic | |||
| Injection: 0.4 mg/mL, available in two pack sizes containing 0.8 mg naloxone hydrochloride in 2 mL or 2.0 mg naloxone hydrochloride in 5 mL |
This is nowhere near a wide-enough array of options, particularly since not all patients respond to these medications and the rate of relapse is still high with all of them. Developing new medications and improved formulations of these existing drugs to enhance compliance, for example with depot formulations or implants, is a top research priority for NIH’s National Institute on Drug Abuse (NIDA).
Moreover, scientific advances in our understanding of the molecular machinery implicated in the brain changes associated with addiction has expanded the array of available medication targets.
There are vast opportunities for small businesses to develop new technologies that can be useful in addressing the opioid epidemic.
In the December issue of Neuropsychopharmacology, NIDA’s Division of Therapeutics and Medical Consequences identified its “ten most wanted” pharmacological mechanisms for OUD medications development, based on the probability of gaining near-term FDA approval for compounds acting on these receptor systems.
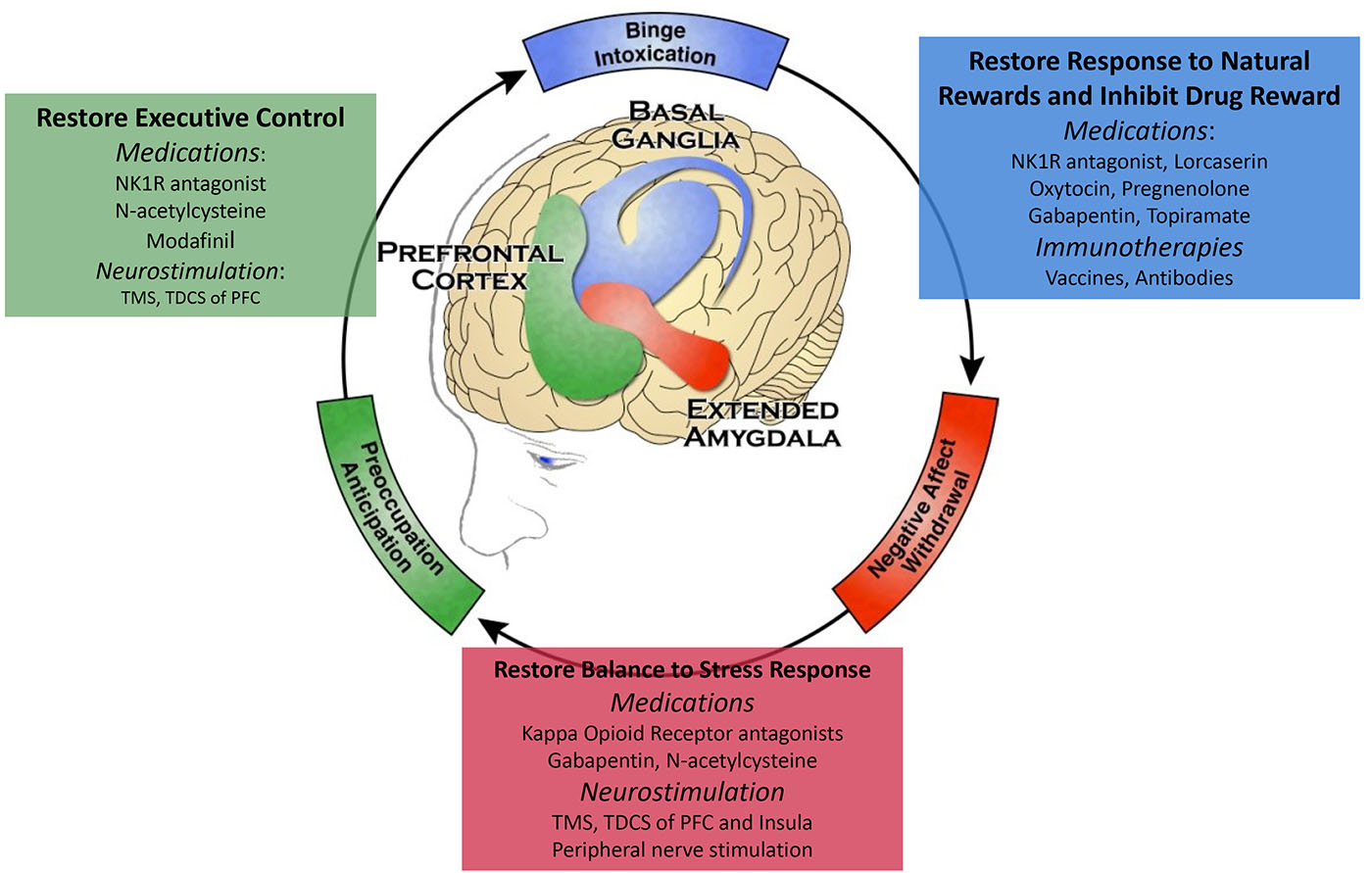
Of particular interest are positive or negative allosteric modulators at several non-μ opioid receptor sites, as these compounds may produce more relevant effects at those receptors than simple agonists or antagonists. In parallel, use of neurostimulation as a strategy to strengthen circuits disrupted by addiction is emerging as an area of great opportunity, which would benefit from innovation in device development (see Figure: “Targets for Addiction Intervention”).
The Focused Opioid Use Disorder Medications Development Project, part of the NIH HEAL (Helping to End Addiction Long-term) Initiative, aims to accelerate the development of new medications and immunotherapies for OUD. It will also facilitate and support development of devices that can be used for OUD treatment by themselves or in combination with MOUD.
Figure: Targets for Addiction Intervention
Examples of potential treatments being evaluated that target distinct components of the main brain regions implicated in the relapsing stages of addiction. NK1R (TACR1) - neurokinin 1; PFC - Prefrontal cortex; TMS - Transcranial magnetic stimulation; TDCS - Transcranial direct-current stimulation
Rethinking the endgame
The increased openness of the FDA to endpoints other than abstinence in the development of medications to treat substance use disorders promises to facilitate success in the approval of new medications and devices. The position was described in a joint publication last year in Science with Janet Woodcock, director of FDA’s Center for Drug Evaluation and Research (CDER).
Alternative endpoints such as clinically meaningful reductions in use (short of cessation), improved sleep, measurable reductions in cravings or withdrawal symptoms, or reduced overdoses for instance, both widen the scope of potential therapeutic targets and make trials easier to conduct.
Another area in need of innovation and of potential interest to industry is that of interventions that target the estimated 12 million Americans who misuse opioids but who do not yet have an opioid use disorder. They too are at risk for overdose, and their opioid misuse constitutes the main risk factor for developing an OUD. Targets for development could include biomarkers that predict risk, devices that can be used to support behavior change or monitor drug misuse, among other innovations.
Eradicating opioid addiction will also require that we restructure how we fund evidence-supported treatments.
There are vast opportunities for small businesses to develop new technologies that can be useful in addressing the opioid epidemic.
Through our Small Business Innovation Research (SBIR) and Small Business Technology Transfer (STTR) programs, NIDA works with companies to develop devices and apps that bring evidence-supported interventions to consumers.
These include mobile apps that deliver or support OUD treatment, predictive tools to help hospital staff detect early diversion of opioids, products to inactivate unused opioid prescriptions, and devices to detect opioid overdoses, among many others.
NIDA has already helped companies successfully develop mobile health technologies to enable physicians to easily monitor and reward patient adherence to their drug regimens, apps that deliver behavioral interventions like cognitive behavioral therapy and link users to peers for support, technologies that expedite linkage to inpatient treatment, and new models of delivering MOUD that utilize digital health to reduce doctor visits and reduce costs, among many others.
Eradicating opioid addiction will also require that we restructure how we fund evidence-supported treatments. The NASEM report recommends developing value-based arrangements in which insurers reimburse providers at a higher rate when a patient is successfully maintained on medications long-term. This pay-for-performance model has already been tried with the antagonist MOUD extended-release naltrexone (see “New Shops for Value-Based Deals”).
Yet despite parity laws, large numbers of treatment programs lack the infrastructure to charge insurance companies and only accept cash payment. Technologies and systems that could facilitate insurance charges are another ripe area for investment.
Addressing the socioeconomic factors that predispose individuals to opioid use and opioid addiction and that interfere with recovery also need to be part of our prevention, treatment and recovery mindset, and here too there may be untold opportunities for new businesses that could target predisposing factors like homelessness, lack of education and difficulty finding a job.
It is equally important to develop safer and more effective treatments for pain that could minimize the use of opioids while not jeopardizing the care of patients, especially those suffering from chronic pain conditions. There are currently 50 million people in America with some form of chronic pain. Some of them are desperate, as the healthcare response to the opioid crisis has been to clamp down on prescribing, leaving some patients who have legitimate need for opioid medications without treatment.
This clinical need, coupled with progress in our understanding of pain processing, also offers many opportunities for pharma and the biomedical device industry.
Not only do we need safer and more effective analgesics, but there is a need for biomarkers that can be used to objectively assess pain and tailor personalized interventions for its treatment. The NIH, via the HEAL initiative, is also prioritizing solutions for the treatment of pain that are effective and non-addictive.
The sky is the limit for creative businesses applying innovative scientific solutions to preventing opioid misuse, helping physicians screen for misuse or substance use disorders, retaining patients in treatment, preventing relapse, and accessing and delivering the care that people with opioid addiction need to successfully recover.
Nora Volkow is director of NIH’s National Institute on Drug Abuse. Comments and ideas are welcome--please send to websubmission@mail.nih.gov.
Guest commentaries reflect the views of the author, not necessarily those of BioCentury.
Companies and Institutions Mentioned
National Academies of Sciences, Engineering and Medicine (NASEM), Washington, D.C.
National Institute on Drug Abuse (NIDA), Bethesda, Md.
National Institutes of Health (NIH), Bethesda, Md.
U.S. Food and Drug Administration (FDA), Silver Spring, Md.
BCIQ Target Profiles